|
|||||||||
|
|
|||||||||
|
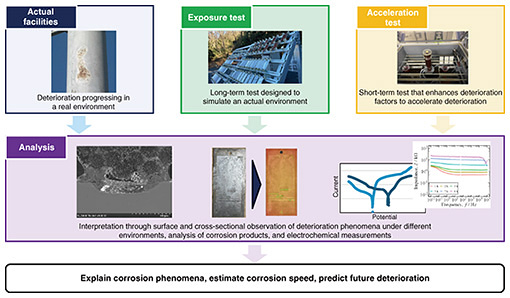
Feature Articles: Research and Development toward Sustainable Infrastructure Prediction of Corrosion Deterioration Based on State of Individual Facilities for Appropriate Maintenance of Communication InfrastructureAbstractCorrectly understanding corrosion behavior under individual environments and predicting deterioration is important in maintaining a communication infrastructure in a sustainable manner. Targeting metal components installed inside maintenance holes, this article introduces efforts for predicting the deterioration of metal inside a maintenance hole by investigating acceleration tests that simulate the maintenance-hole environment. Keywords: metal corrosion, acceleration test, maintenance hole 1. Social infrastructure maintenance and corrosionThe structures that make up the social infrastructure are many and varied. In addition to streel structures composed mainly of iron, such as towers, bridges, and plant facilities, there are reinforced concrete structures, such as building frames, bridge piers, and retaining walls. The functionality of these long and massive structures is maintained not only with major components but also with a variety of metallic components, fittings, etc. In addition to high mechanical properties, iron excels in economic efficiency and workability while having good recyclability. Because of these features, iron has become widely used as a material supporting the social infrastructure regardless of structure size. Under long-term usage, however, there are concerns that materials made of iron will deteriorate due to corrosion. Ongoing corrosion cannot only reduce the safety and functionality of a structure but also increase the frequency of repairs and renovations, thus increasing the social and economic burden. Developing advanced technologies for correctly understanding corrosion behavior and suppressing/predicting deterioration is important. Such technologies can provide a platform for supporting a safe and reliable social infrastructure while simultaneously helping reduce the maintenance expenses of company-owned facilities. Against this background, it is important to focus on individual components making up the social infrastructure and understand their corrosion behavior. 2. Flow of evaluating corrosion behaviorTo evaluate corrosion behavior, it is important to analyze actual facilities and investigate exposure tests and acceleration tests*1. The analysis of actual facilities will clarify the environmental conditions in which equipment, structures, etc., are actually placed and determine the form and distribution of the corrosion. This will enable exposure tests and acceleration tests to be designed on the basis of the actual environment and state of corrosion. An exposure test is used to identify some of the varied types of disturbances that occur in actual facilities, making it easier to understand the progression of corrosion. It features the ability to continuously observe corrosion behavior from the initial state of environment exposure. However, a long test period is required to conduct an evaluation. An acceleration test, on the other hand, can accelerate deterioration by amplifying some of the corrosion factors in actual facilities so that influencing factors can be extracted in a short time and their impact evaluated. However, care must be taken to avoid discrepancies with the actual environment. In short, conducting a multifaceted evaluation by combining surface analysis and the analysis of corrosion products along with cross-sectional observations, electrochemical measurements, etc. opens the door to clarifying the corrosion mechanism, determining the speed of corrosion, tracking the deterioration process, and predicting deterioration. The flow of corrosion evaluation is shown in Fig. 1.
The outside communications infrastructure owned by NTT includes communication buildings, tunnels, underground conduits, maintenance holes, and utility poles. Appropriate maintenance had been carried out by conducting facility surveys, exposure tests, and acceleration tests for each type of facility and predicting deterioration. This article focuses on an example of studying acceleration tests targeting metal components installed inside maintenance holes.
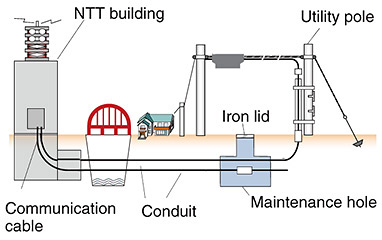
3. Maintenance-hole maintenanceIron lids that can be seen on streets and sidewalks in the city are often called maintenance holes, but it would be more accurate to call the structure under such an iron lid a maintenance hole. Maintenance holes may serve as inlets and outlets for the water supply and sewage and may also function as connectors of communication facilities and as workspace for maintaining those facilities. NTT owns about 680,000 maintenance holes for communication purposes throughout Japan. As shown in Fig. 2, NTT communication cables pass through maintenance holes before being erected on aerial facilities such as utility poles and strung to user homes. These communication cables support a communication infrastructure that is indispensable for daily life, so it is essential that they are operating stably at all times.
Inspections are currently conducted using a method for periodically recording the state within a maintenance-hole space using a 360-degree camera to check the deterioration condition. Going forward, it will be necessary to make inspection operations even more efficient to deal with a decrease in maintenance personnel, hold down maintenance expenses, and maintain a safe and secure communications infrastructure. With this in mind, NTT will use the results of deterioration prediction to conduct early inspections of maintenance holes having a high risk of deterioration while extending the inspection period of such facilities with a low risk of deterioration. We at NTT Device Technology Laboratories will undertake the development of well-balanced maintenance techniques corresponding to the state of individual facilities. 4. Role of fittings inside maintenance holeIn addition to managing the maintenance-hole frame, maintenance-hole maintenance targets the inspection and repair of ducts that connect conduits and of metallic components that pull and bear communication cables. To make inspections more efficient, each component’s manner of deterioration must be understood, and the means of making maintenance more efficient must be studied. We are focusing on metal fittings installed inside a maintenance hole for bearing cables. Inside a maintenance hole, a laid communication cable is supported by fittings attached to a concrete wall, which makes it easy for a worker to connect cables [1]. A communication cable and fitting are also fixed by a string to prevent the cable from moving or falling. 5. Corrosion environment inside maintenance holeInside a maintenance hole, rainwater or groundwater flowing in from gaps in the iron lid or from ducts can become stagnant, forming an environment in which metal can easily corrode. If a fitting should deteriorate and become damaged due to corrosion, there are concerns that the communication cable will drop, adversely affecting communications. There is therefore a need for appropriately maintaining and managing such fittings by understanding the manner in which they deteriorate. However, the environment within maintenance holes is not sufficiently understood, making it difficult to clarify the corrosion behavior of fittings. Since the interior of a maintenance hole is a nearly sealed space, the flowing in of water from the outside can cause the space inside the maintenance hole to become a high-humidity environment near a humidity of 100%. If a large amount of water is flowing in, the fitting may become submerged in stagnant water. It has been reported that a fitting in stagnant water can more easily corrode the closer it is to the water surface [2]. The environment of such a fitting therefore differs according to the level of stagnant water, so the manner of corrosion is also expected to change for each type of environment. The level of stagnant water can vary according to the amount of water inflow/outflow, so a fitting near the water surface can be exposed to an environment in which high-humidity conditions and submerged conditions alternate (air/solution alternating environment). As shown in Fig. 3, an example of local corrosion occurring at the position where the cable is assumed to have been secured by a string has been confirmed from actual facilities, so there is a need to clarify such singular corrosion behavior at the part of a fitting in contact with the string. We are therefore focusing on corrosion near the part of a fitting in contact with a string in an air/solution alternating environment in which corrosion deterioration is thought to be the most significant within a maintenance hole. Our aim is to predict deterioration of a fitting by estimating long-term deterioration by acceleration testing and clarifying corrosion behavior by electrochemical measurements.
5.1 Estimating long-term deterioration by acceleration testing5.1.1 Acceleration test methodsWe used two types of samples in our acceleration tests: steel plate hot-rolled commercial (SPHC) steel plates and zinc-coated steel plates achieved by subjecting SPHC to hot-dip galvanizing. To align the corrosion range of each, we masked the back and four sides of each sample, and to simulate corrosion at a section in contact with a string, we tied a string six times around the exposed part of a steel plate. While there are multiple methods and conditions in acceleration testing, we selected three tests for this study: salt spray test (SST), cyclic corrosion test (CCT), and air/solution alternating test. The SST and CCT are standard tests that are widely used to evaluate corrosion in air. As the name implies, the SST is used to execute continuous salt spraying (5% sodium chloride), while the CCT cycles through two hours of salt spraying, four hours of drying (temperature: 60°C, humidity: 20%), and two hours of wetting (temperature: 50°C, humidity: 95%). As described above, corrosion behavior in a maintenance-hole environment differs from that in an air environment, so we decided to include an air/solution alternating test as an acceleration test that can better simulate corrosion in a maintenance-hole environment. An air/solution alternating test alternates between a state in water and a state in air. In this study, we repeated a cycle of 144 hours in a water state and 24 hours in an air state. The solution used in the water state was a 5% sodium chloride solution, and the environment for the air state was set to 50°C with 95% humidity, the same as the wetting state. The test times for each acceleration test were 480 hours for the SST and CCT and 672 hours for the air/solution alternating test. After completion of an acceleration test, we removed the rust attached to the sample and determined the amount of corrosion thinning using a method based on a Japanese Industrial Standards (JIS) standard (JIS H 8502). After drying a sample with rust removed, we measured the unevenness of its surface using a three-dimensional shape measuring machine. 5.1.2 Acceleration test resultsRust was removed from post-acceleration-testing samples (with string). Contour diagrams*2 showing the surface unevenness of those samples are shown in Fig. 4: an external view of a sample (with string) before acceleration testing is shown in (a), SPHC contour diagram after 480 hours of the CCT is shown in (b), galvanized-plate contour diagram after 480 hours of the CCT is shown in (c), SPHC contour diagram after 480 hours of the SST is shown in (d), and SPHC contour diagram after 672 hours of the air/solution alternating test is shown in (e). The contour diagrams are shown with a color scale in which the masked location at the top of the sample was taken to be 0 µm. A decrease in plate thickness due to corrosion thinning at unmasked locations was apparent. From Figs. 4(b) and (c), a decrease in thickness was greater for SPHC than galvanized steel plates. From Figs. 4(b) and (d), while most of the string-contact section in the CCT and SST samples did not decrease in thickness more than that of the exposed section, a decrease in thickness equal to or greater than that of the exposed section did occur directly under the string at the top of the string-contact section. Finally, from Fig. 4(e), a decrease in thickness was greater in the string-contact section than in the exposed section in the air/solution alternating test sample. In contrast to the CCT and SST samples, a decrease in thickness was observed in the sample directly under all six string windings in the string-contact section and not just at the top of this section.
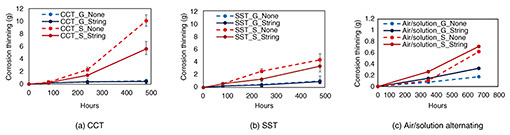
The change in overall corrosion thinning of each sample is shown in Fig. 5. The values in the graphs plot the average of multiple samples (CCT and SST: N = 4, air/solution alternating: N = 3) after acceleration testing. These results indicate that the amount of overall corrosion thinning tended to be greater for the SPHC than galvanized samples for all three acceleration tests. Overall corrosion thinning tended to be greater for samples during the CCT compared with the SST and greater for samples during the SST compared with the air/solution alternating test. Overall corrosion thinning with/without the string tended to be greater for samples without the string during the CCT and SST for both SPHC and galvanized samples, but for the air/solution alternating test, it tended to be greater for samples with the string.
5.2 Clarifying corrosion behavior by electrochemical measurementsMetal corrosion is an electrochemical reaction consisting of two simultaneous reactions, one in which the metal loses electrons (anode reaction) and the other in which electrons are received in a solution (cathode reaction). Electrochemical measurements are therefore effective for investigating the progression and speed of corrosion. These measurements can also be used to evaluate the initial behavior of corrosion by capturing minute amounts of current flowing in conjunction with that corrosion. A three-electrode system is generally used consisting of a working electrode as the measurement target, reference electrode that provides a base potential, and counter electrode to drive the current necessary for making the measurements. The speed of corrosion and reaction characteristics can be quantitatively understood by measuring current while controlling potential using a potentiostat. Typical electrochemical measurement methods used for evaluating corrosion include natural potential measurement, polarization curve measurement, polarization resistance, and electrochemical impedance spectroscopy (EIS). EIS can separate the effects of charge movement, diffusion, and corrosion-produced film in evaluating corrosion by measuring responses while varying the frequency of an alternating-current signal. Another key feature of EIS is that it can track the corrosion process over time, changes in the film, etc. with nearly no change in the state of the sample. We are developing a technique for applying EIS to metallic samples in a state in contact with a string to quantitatively evaluate corrosion speed in a process in which the corrosion environment moves from water to air after which moisture on the metal surface evaporates. We have thus far confirmed that corrosion speed increases in such a moisture-decreasing process and consider the main factor to be local corrosion at the string-contact section, as observed from acceleration tests (studies are progressing in this regard).
6. ConclusionWe were not able to reproduce local corrosion directly under a string during the CCT and SST. We consider the reason for this to be that the string prevents the sprayed saline water from contacting the steel plate where it comes in contact with the string. However, for the section directly under the uppermost part of the string-contact section toward the top of the sample, the saline water flowing from the top of the sample is absorbed by the string, which we consider be the reason that local corrosion occurred. The fact that corrosion progresses further during the CCT than the SST suggests that local corrosion of a fitting within a maintenance hole can easily occur in an environment that alternates between high-humidity conditions and submerged conditions in which moisture is sufficiently absorbed by the string. We were able, however, to reproduce local corrosion directly under the string in the air/solution alternating test. We consider the reason for this to be that since the cycle interval in the air/solution alternating test is long, the progression of deterioration by corrosion was somewhat gradual at unmasked locations. In the section in contact with the string, however, a sufficient amount of moisture was absorbed throughout the string in a submerged state while drying occurred in the air state causing local corrosion to progress directly under the string. As shown in Fig. 4(e), the amount of corrosion thinning was greater directly under the string than at locations with no string attached. In short, it is possible to reproduce local corrosion at the section of a fitting in contact with a string by conducting the air/solution alternating test. References
|
|||||||||