|
|||||||
|
|
|||||||
|
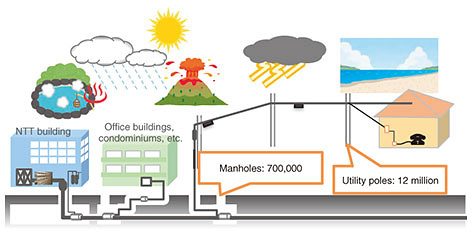
Practical Field Information about Telecommunication Technologies Vol. 19, No. 6, pp. 110–114, June 2021. https://doi.org/10.53829/ntr202106pf1 Sulfur Damage and Its Countermeasures in Telecommunication Equipment and FacilitiesAbstractThis article introduces cases of corrosion caused by hydrogen sulfide in telecommunication equipment and facilities and countermeasures against it. This is the sixty-fourth article in a series on telecommunication technologies. Keywords: hydrogen sulfide, sulfur damage, corrosion 1. IntroductionTelecommunication facilities, such as utility poles (Fig. 1), are installed throughout Japan and exposed to various natural phenomena. These facilities are composed of various materials, such as metals, plastic, and concrete, and gradually deteriorate due to corrosion and abrasion caused by the surrounding environment. In coastal areas, for example, salt damage—which is corrosion caused by particles of sea salt being blown by strong winds and landing onto telecommunication facilities containing metal—is a well-known phenomenon. In Japan, salt damage is unavoidable because of the long coastline. Japan is also a volcanic country, having numerous hot springs. In many of these areas, hydrogen sulfide (H2S), which is a gas containing sulfur, is emitted from these hot springs. Since H2S is highly corrosive, it can deteriorate not only outdoor telecommunication facilities but also indoor ones. This type of corrosion is called sulfur damage. Where sulfur damage is severe, terminal equipment fails every few months, so maintenance operations to fix such failures are burdensome. In response to on-site requests, the Technical Assistance and Support Center (TASC), NTT EAST, has investigated instances of sulfur damage on outdoor and indoor equipment and facilities. This article introduces the mechanism of sulfur damage, actual cases of sulfur damage to telecommunication equipment and facilities, and countermeasures against it.
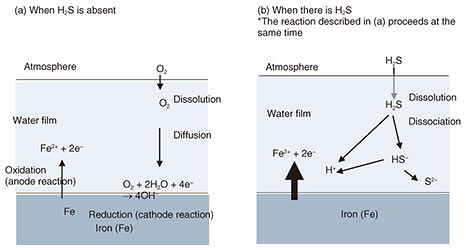
2. Mechanism and actual cases of sulfur damage in telecommunication equipment and facilities2.1 Mechanism of sulfur damageThe corrosion reaction that causes sulfur damage occurs when a water film is formed on the surface of a material by condensation or moisture adsorption. As shown in Fig. 2(a), when H2S is absent, the corrosion of iron is caused by oxygen in the atmosphere dissolving in the water film, followed by reduction of the dissolved oxygen on the surface of the iron and simultaneous dissolution of the iron. When sea salt is also dissolved in the water film, the corrosion rate of iron will increase due to the increased electrical conductivity of the water film. When there is H2S present (Fig. 2(b)) in addition to the reactions illustrated in Fig. 2(a), the H2S dissolves and dissociates in the water film to produce H+ ions, which results in an acidic environment in the water film. This acidity accelerates the dissolution of iron and results in a high corrosion rate.
2.2 Actual cases of sulfur damage2.2.1 Sulfur damage to outdoor facilitiesExamples of sulfur damage to outdoor facilities at a hot-spring area are shown in Fig. 3. Significant rusting (reddish brown) was found on the metal bands at the lower part of the guy-line rod and scaffolding bolts on the utility pole. Thinning can be seen in the central part of the guy-line rod, so the rod may break when corrosion progresses. Although this type of corrosion can be caused by either salt or sulfur damage, since this hot-spring area is located in a mountainous area far from the coast, the effect of salt damage is considered small. Moreover, measurements of gas concentration at the locations shown in Figs. 3(a) and (b) detected H2S at 1.5 ppm (parts per million) and 0.9 ppm, respectively. The above corrosion phenomena are thus considered to be sulfur damage, the main factor of which is H2S. The regulation value for atmospheric concentration of H2S stipulated by the Offensive Odor Control Law is 0.06 to 0.2 ppm [1], and the Industrial Safety and Health Act stipulates that the concentration of H2S should be less than 1 ppm to ensure a safe working environment [2]. Accordingly, the measured H2S concentrations (1.5 and 0.9 ppm) at these facilities indicate that H2S exists at high concentrations at those locations.
2.2.2 Sulfur damage to indoor equipmentThe corrosion rate of metals in an indoor environment is generally lower than that in an outdoor environment because the intrusion of sea-salt particles and H2S is hindered. However, if indoor spaces (rooms, etc.) are not sufficiently sealed, corrosion may occur due to the intrusion of sea-salt particles or H2S. Since H2S is a gas, it is difficult to completely prevent it from entering indoor spaces. TASC has conducted a number of field investigations of indoor equipment after receiving requests for technical consultations from service personnel. An example of sulfur damage to indoor equipment is shown in Fig. 4, which illustrates sources of H2S (Fig. 4(a)), examples of metal corrosion in indoor equipment (Fig. 4(b)), and micrographs of a circuit board inside a piece of terminal equipment (Fig. 4(c)). In this example, the concentrations of H2S near the hot-spring-water vat and fumaroles, which are thought to be the sources of H2S, were 0.1 and 0.9 ppm, respectively. It was also confirmed that metal fixtures, such as windows and faucets, were significantly corroded in the indoor environment, and it is considered that the corrosion is caused by the intrusion of H2S from outdoors.
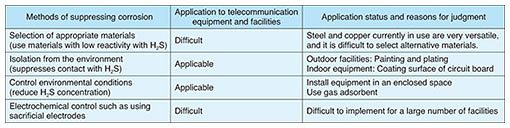
In the other case investigated by TASC, a ppb (parts per billion, where 1 ppb = 1/1000 ppm) level of H2S concentration was detected in indoor environments. From the results of scanning electron microscopy - energy dispersive X-ray spectrometry (SEM-EDS) analysis, the main component of the corrosion depicted in the figure was sulfide, which indicates that a reaction between the metal and H2S had occurred. Black corrosion was found on copper wiring and around through holes by microscopic observation of a circuit board collected from a faulty piece of terminal equipment installed in the same indoor space. SEM-EDS analysis of this black corrosion revealed that it contained sulfur in a similar manner to the indoor metal fixtures described above, and X-ray-diffraction analysis of the corrosion identified the main component as copper sulfide. When the copper wiring pattern on a circuit board is corroded by H2S and becomes copper sulfide, it grows on the circuit board and connects to adjacent wiring, through holes, etc. It is thought that since the copper sulfide is conductive, a short circuit occurred between the wiring and through holes, which should not be connected, causing the equipment failure. 3. Countermeasures against sulfur damage3.1 Basic requirements for countermeasures against sulfur damageAs the installation standard for terminal equipment, JEITA IT-1004 (Standard for Operating Conditions of Industrial Computer/Control System) of the Japan Electronics and Information Technology Industries Association [3] recommends controlling H2S concentration to 3 ppb or less. There are several methods of suppressing corrosion caused by H2S, as listed in Table 1. Taking costs and other factors into consideration, the basic requirements of countermeasures against sulfur damage to telecommunication equipment and facilities were determined as suppressing contact with H2S and reducing H2S concentration.
3.2 Countermeasures to suppress corrosion due to sulfur outdoorsSince concentration of H2S is higher outdoors than indoors, namely, at the ppm level, countermeasures are required to provide higher corrosion protection performance. To meet this requirement, it is necessary to ensure that outdoor facilities and equipment are prevented from coming into contact with H2S. One way to prevent contact is to coat the surface of the base metal with a paint that has stronger adhesion. By increasing the adhesion between the paint coating and base metal, it is possible to suppress (i) direct contact between H2S and the metal and (ii) the adhesion of moisture (water film) containing H2S to the metal. Therefore, it is possible to reduce the occurrence of sulfur damage. An example of metal bands coated with a powder coating, which has stronger adhesion and higher corrosion resistance than conventional coatings, attached to a concrete utility pole is shown in Fig. 5.
3.3 Countermeasures to suppress corrosion due to sulfur indoorsAn example of a countermeasure against sulfur damage to indoor telecommunication equipment is shown in Fig. 6. The concentration of H2S can be reduced by installing the terminal equipment inside a sealable acrylic box and placing H2S adsorbent inside. In fact, a customer who had been experiencing failures of terminal equipment every two months or so has been free of failures for more than 10 months after starting to use an acrylic box.
4. ConclusionEffective countermeasures to protect telecommunication equipment from sulfur damage is to (i) prevent the equipment from coming into contact with H2S and (ii) reduce the concentration of H2S in the vicinity of the equipment. Housing indoor equipment in acrylic boxes containing H2S adsorbent has been implemented. For outdoor facilities, a powder coating, which provides better corrosion protection than conventional zinc plating, can be used. Applying these countermeasures will extend the life of telecommunication equipment and facilities. TASC will continue to promote technical-cooperation activities to solve problems in the field, such as those related to degradation of equipment and facilities due to sulfur and salt damage. Through these activities, we will contribute to improving the quality and reliability of telecommunication services. References
|
|||||||